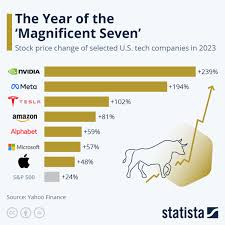
by: moneycontrol.com
Asia's Stock Markets Face Headwinds: AI Bubble, Policy Divergence & Geopolitical Risks
by: Ghanaweb.com
Ghana Minister Proposes Investing Excess Liquidity into Venture Capital & Stock Market

Zevra Therapeutics: High-Risk, High-Reward Rare Disease Play?





Zevra Therapeutics: A High-Risk, High-Reward Play in Rare Disease Treatment?
Zevra Therapeutics (ZEVRA) is a small-cap biopharmaceutical company attracting attention for its potential within the ultra-rare disease market. According to a recent Seeking Alpha analysis, the stock presents an "asymmetric risk-reward setup," meaning the upside potential significantly outweighs the downside risks – if certain key milestones are achieved. However, this opportunity comes with substantial caveats and dependencies on clinical trial success and regulatory approval. This article will break down Zevra's core assets, the rationale behind the bullish thesis, the significant risks involved, and ultimately, whether it’s a worthwhile investment for risk-tolerant investors.
The Core Asset: Taviglucer (Zavesca) & Beyond
At the heart of Zevra's strategy is Taviglucer, marketed as Zavesca in Europe. Taviglucer is an enzyme replacement therapy designed to treat Gaucher disease type 2 (GD2), a devastating and ultra-rare lysosomal storage disorder characterized by severe neurological dysfunction and typically leading to death in early childhood. It's currently approved in Europe but not in the United States, representing Zevra’s primary near-term catalyst. The Seeking Alpha article highlights that Taviglucer has demonstrated a survival benefit for GD2 patients in Europe, which is crucial for potential FDA approval.
Beyond Taviglucer, Zevra also holds rights to other therapies targeting rare diseases, including Adagrasib (a KRAS G12C inhibitor) and potentially others through its licensing agreements. While these represent future possibilities, the immediate focus remains on Taviglucer's US approval. The company’s strategy is centered around acquiring or in-licensing promising treatments for underserved rare disease populations, leveraging their commercialization expertise to bring them to market.
The Bullish Thesis: A Significant Untapped Market & Potential FDA Approval
The bullish argument for Zevra rests on several pillars:
- Large Unmet Need: GD2 is incredibly rare – estimates suggest only around 100-200 patients in the US. While this small patient population might seem insignificant, it translates to a potentially high price per treatment and substantial revenue generation if Zavesca gains FDA approval. The Seeking Alpha article emphasizes that even conservative estimates of market penetration could lead to significant sales.
- Positive European Data: The existing data from Europe showing improved survival rates for GD2 patients treated with Taviglucer is compelling. This provides a strong foundation for the US regulatory submission and increases the likelihood of approval, although it's not guaranteed (more on that below).
- Orphan Drug Designation & Incentives: Zavesca has received Orphan Drug Designation in the United States, granting Zevra various incentives including market exclusivity (seven years post-approval) and tax credits for clinical development. This significantly reduces the risk associated with bringing a rare disease drug to market.
- Potential for Expansion: While initially focused on GD2, there's potential to explore Taviglucer’s efficacy in other Gaucher variants or even related lysosomal storage disorders. This could broaden the addressable market considerably.
- Attractive Valuation: The Seeking Alpha analysis suggests that Zevra is currently undervalued by the market, largely due to the risks associated with the FDA approval process. The potential upside, if successful, would significantly outweigh the current share price.
The Risks: A Minefield of Regulatory and Clinical Hurdles
Despite the compelling bullish case, significant risks loom large for Zevra investors:
- FDA Approval Uncertainty: This is the critical risk. The FDA has stringent approval criteria, and while European data is helpful, it's not a guarantee of US approval. The Seeking Alpha article points out that the FDA may require additional clinical trials specifically designed to meet US regulatory standards. This could delay or even derail the approval process.
- Clinical Trial Design & Execution: Zevra plans to initiate a pivotal Phase 3 trial for Taviglucer in GD2 patients. The success of this trial is paramount. Poorly designed or executed trials can lead to negative results and jeopardize FDA approval. The article highlights that patient recruitment, given the rarity of the disease, will be a significant challenge.
- Commercialization Challenges: Even with FDA approval, successfully commercializing Zavesca in the US will require a robust sales and marketing infrastructure. Zevra needs to effectively reach specialists treating GD2 patients and navigate complex reimbursement hurdles. The Seeking Alpha article questions whether Zevra has sufficient resources for this undertaking.
- Competition: While GD2 is ultra-rare, potential competitors could emerge with alternative therapies or improved treatments. This would erode Zevra’s market share and pricing power.
- Financial Risk: As a small biopharmaceutical company, Zevra relies heavily on external financing. Dilution through future stock offerings is a possibility, which could negatively impact existing shareholders. The Seeking Alpha article notes the company's current cash runway and potential need for additional funding.
- Adagrasib Development & Commercialization: While Adagrasib represents a longer-term opportunity, its development and commercialization are also subject to significant risks, including clinical trial failures and competition from other KRAS inhibitors.
Conclusion: A Speculative Investment with High Potential – and High Risk
Zevra Therapeutics presents a compelling but highly speculative investment opportunity. The potential reward—FDA approval of Taviglucer for GD2 and subsequent commercial success—is substantial. However, the risks are equally significant, primarily centered around FDA approval uncertainty and clinical trial execution. The "asymmetric risk-reward setup" described in the Seeking Alpha article is real, but it’s crucial to understand that this opportunity isn't suitable for risk-averse investors. A thorough understanding of the disease, the regulatory landscape, Zevra’s financial position, and the competitive environment is essential before considering an investment. Investors should closely monitor the progress of the Phase 3 trial and any updates from the FDA. Ultimately, Zevra's fate hinges on its ability to navigate the complex path towards bringing a life-saving treatment to patients with GD2 in the United States.
Read the Full Seeking Alpha Article at:
https://seekingalpha.com/article/4856967-zevra-therapeutics-stock-asymmetric-risk-reward-setup-in-ultra-rare-disease-market
on: Thu, Dec 18th 2025
by: Seeking Alpha
Day One Pharmaceuticals Stock Soars Over 30% on Breakthrough Oral GLP-1 Data

on: Thu, Sep 11th 2025
by: Seeking Alpha
Ionis Stock: Renewed Assault On $100.00 Per Share (NASDAQ:IONS)

on: Mon, Mar 03rd 2025
by: MSN
Is Kiniksa Pharmaceuticals International, plc (KNSA) the Best Small Cap Pharma Stocks to Buy Now?

on: Thu, Dec 04th 2025
by: The Motley Fool

on: Fri, Nov 21st 2025
by: The Motley Fool

on: Thu, Jul 31st 2025
by: Seeking Alpha
Dyadic International Announces $10 Million Public Stock Offering

on: Tue, Jul 29th 2025
by: Forbes

on: Tue, Jan 07th 2025
by: MSN
Here's Why MannKind Corporation (NASDAQ:MNKD) Is Among the Best Diabetes Stocks to Buy Under $10

on: Mon, Dec 29th 2025
by: Seeking Alpha
Southpoint Capital Exits $75 Million Stake in Fennec Pharmaceuticals

on: Fri, Dec 19th 2025
by: Seeking Alpha
Insmed's Pulmonary Promise Holds, Yet Profitability Remains Elusive

on: Sat, Dec 13th 2025
by: Seeking Alpha
Eli Lilly's Long-Term Playbook Focuses on Diabetes, Oncology, Immunology

on: Sat, Dec 06th 2025
by: Seeking Alpha
Viking Therapeutics Targets 2026 as Critical Year for Gene-Editing Breakthrough