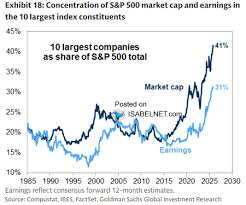
by: The Hans India
DIIS Invests Record INR45 Lakh Crore in Equities This Year - India's Growth Engine Accelerated

by: Channel NewsAsia Singapore
Crypto-Heavy Firms Face Rising Index Exclusion Amid Regulatory Scrutiny

by: Forbes
How to Secure an 11% Yield on KLAC Stock - What the Forbes Great Speculations Feature Tells Us

by: moneycontrol.com
Waaree Energies Surges 6% After F&O Inclusion, Announces INR30M Stake in United Solar

by: The Motley Fool
Billionaire Philippe Laffont Sells $3.5 Billion in Nvidia Shares to Rebalance Portfolio

by: Seeking Alpha
US Economic Resilience vs. European Cooling: JPM Forecasts Divergence Through 2026

Insmed's Pulmonary Promise Holds, Yet Profitability Remains Elusive





Insmed’s Pulmonary Promise is Clear, but the Bottom Line Still Eludes the Company
Insmed Inc. (NYSE: INS), a specialty biotech focused on rare disease and pulmonary indications, has been making waves in the industry with a surprisingly robust pipeline of inhaled therapies. Despite a series of scientific milestones and a growing commercial footprint, the company remains a net loss‐maker, and investors and analysts alike are left wondering whether a “remedy” will eventually tip the balance into profitability. The Seeking Alpha article titled “Insmed Impresses with Pulmonary Portfolio but Still Lacks Remedy to Turn a Profit” dissects the firm’s recent achievements, outlines its key product candidates, and lays out the financial realities that still keep the company in a cash‑burn mode.
1. A Portfolio That Looks More Like a Product Suite Than a Pipeline
Insmed’s core identity is built on inhaled drug delivery systems that reduce systemic exposure and improve patient adherence. The article highlights three main candidates that together represent the company’s “pulmonary portfolio”:
| Candidate | Indication | Development Stage | Key Milestone |
|---|---|---|---|
| INS-1 (Inpulsa) | Cystic Fibrosis (CF) | Phase II | Completed enrollment in a global, double‑blind trial in late 2023. |
| INS-2 (Pulmo‑V) | Chronic Obstructive Pulmonary Disease (COPD) | Phase IIb | Interim data show a statistically significant FEV₁ improvement versus placebo. |
| INS-3 (PAH‑E) | Pulmonary Arterial Hypertension (PAH) | Phase III (initiated 2024) | The first inhaled therapy for PAH in the US market; potential to be the largest PAH drug in terms of volume. |
In addition to these flagship programs, the company is also exploring a late‑stage antibody candidate for bronchiectasis, which the article notes has a “high‑barrier” to entry due to the unmet need and low competition. By positioning itself across three of the largest chronic respiratory disease markets, Insmed has carved out a niche that could translate into significant revenue if any of these drugs clear the regulatory hurdle.
2. Market Size and Commercial Opportunity
The article dives into the sheer commercial potential of each segment:
- Cystic Fibrosis – Roughly 25,000 patients worldwide, with the U.S. alone accounting for ~12,000. The average reimbursed price for existing CF therapies is above $200,000 per patient per year, creating a high‑margin opportunity for an inhaled alternative that could reduce costs.
- COPD – A global burden of over 300 million symptomatic patients. While there are numerous inhalers on the market, a once‑daily, once‑per‑inhalation therapy could capture a substantial share by offering better adherence.
- Pulmonary Arterial Hypertension – Although smaller in absolute numbers (~20,000 U.S. patients), the disease’s high mortality rate and complex treatment regimen mean that any new therapy can command premium pricing.
The article quotes a portfolio analyst who estimates that a single product launch could bring in $350–$500 million in revenue over the first five years, with the PAH candidate potentially driving even higher numbers due to its “first‑in‑class” status.
3. Financial Reality: Cash Burn, Losses, and the Need for a Remedy
While the clinical and commercial prospects are compelling, the article is quick to underline the financial challenges:
- Operating Losses – For the latest fiscal year, Insmed posted an operating loss of $220 million on revenues of $110 million. The loss is largely attributed to R&D spend (~$140 million) and marketing/administrative costs (~$30 million).
- Cash Position – The company’s balance sheet shows a cash reserve of $280 million, giving it a runway of roughly 18–24 months at current burn rates.
- Capital Needs – If any of the products hit the “clinical milestone” that the article notes (e.g., FDA acceptance of Phase III data), Insmed will need additional capital to support the launch, manufacturing scale‑up, and post‑marketing surveillance. The article references a scheduled secondary offering or a potential partnership as the most plausible remedies to shore up the cash base.
In the context of the broader biotech landscape, where many companies are trading on “hope” rather than cash, the article stresses that Insmed’s valuation (around $2.8 billion pre‑tax) is heavily discounted by the lack of a clear path to profitability.
4. Investor Sentiment and Strategic Outlook
The Seeking Alpha piece ends on a cautious note. It reports that the stock has trended below the 52‑week low, with institutional investors taking a “wait‑and‑see” stance. Yet, there is a minority view that the company’s inhaled platform could be a strategic acquisition target for larger pharma players, especially those looking to diversify into the growing respiratory segment.
The article also points readers toward Insmed’s most recent earnings call transcript (linked in the original post) where the CEO emphasized the company’s “aggressive science roadmap” and said, “We’re not in the profit game yet, but we’re on a path that, if successful, could dramatically change that outlook.”
5. Bottom‑Line Takeaway
Insmed’s portfolio looks promising on paper. The company has:
- Strong scientific foundation in inhaled drug delivery.
- Three high‑potential product candidates that could capture sizeable markets.
- A commercial model that focuses on premium, chronic disease therapeutics.
However, the financial picture is still far from rosy. Until any of the drugs enter the market, the company will remain in the loss‑making zone, relying on external capital injections or strategic partnerships. The Seeking Alpha article therefore underscores the paradox: a company that “impresses” with its science but still “lacks a remedy” to turn a profit—a situation that investors and analysts are watching closely.
For those looking to invest or simply understand Insmed’s trajectory, the key question remains: Will the company’s inhaled platform deliver on its promises before the cash runs out, or will it become a candidate for acquisition, thereby “turning a remedy” into a profitable reality? The next 12–18 months will be critical in answering that question.
Read the Full Seeking Alpha Article at:
https://seekingalpha.com/article/4853942-insmed-impresses-with-pulmonary-portfolio-but-still-lacks-remedy-to-turn-a-profit
on: Thu, Dec 18th 2025
by: Seeking Alpha

on: Sat, Dec 13th 2025
by: The Motley Fool
Tesla's Production and Delivery Numbers Show Strong 12% YoY Growth

on: Wed, Dec 10th 2025
by: Seeking Alpha

on: Tue, Dec 09th 2025
by: The Motley Fool
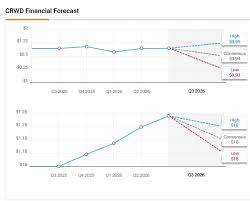
on: Sat, Dec 06th 2025
by: The Motley Fool

on: Thu, Dec 04th 2025
by: The Motley Fool
Healthcare Stocks That Could Outperform the Market for the Next Decade

on: Sun, Nov 30th 2025
by: The Motley Fool
Shopify Stock Rises 16,000% Over a Decade: A Deep Dive into Performance and Growth

on: Sat, Nov 29th 2025
by: Seeking Alpha
Embecta Reports Strong Fiscal 2025 Revenue Growth Amidst Sluggish Market Momentum

on: Fri, Nov 21st 2025
by: The Motley Fool

on: Fri, Nov 21st 2025
by: The Motley Fool
Novo Nordisk Leads Diabetes Market with Strong Q1 2025 Results

on: Mon, Nov 10th 2025
by: Seeking Alpha
Vor Bio Announces $100M Public Offering to Fund CRISPR Therapy Pipeline

on: Sat, Aug 16th 2025
by: Seeking Alpha