[ Sat, Nov 29th 2025 ]: investorplace.com
AI Boom Opens New Pathways: The Backdoor Route to Public Markets

[ Sat, Nov 29th 2025 ]: The Wall Street Journal
Teenagers Turning Their Pennies Into a Future Home and a Retirement Nest Egg

[ Sat, Nov 29th 2025 ]: MoneyWeek

[ Sat, Nov 29th 2025 ]: 24/7 Wall St.
Alphabet Shares Hit New All-Time Highs After NVIDIA H100 Surpasses Google TPUs

[ Sat, Nov 29th 2025 ]: AOL

[ Sat, Nov 29th 2025 ]: CNBC

[ Sat, Nov 29th 2025 ]: CNBC
Alphabet Tops Overbought List in CNBC's 2025 Market Snapshot

[ Sat, Nov 29th 2025 ]: The Globe and Mail
Housing Prices Soar While Young Canadians Turn to Stock Market

[ Sat, Nov 29th 2025 ]: Business Today
HDFC Bank Eyes 18% Profit Surge Amid Strong Loan Growth and Digital Initiatives

[ Sat, Nov 29th 2025 ]: Seeking Alpha

[ Sat, Nov 29th 2025 ]: The Motley Fool

[ Sat, Nov 29th 2025 ]: Seeking Alpha
Alphabet Achieves Resounding Victory in Competitive Tech Landscape

[ Sat, Nov 29th 2025 ]: Business Today
Small-Cap Funds in 2025: How Much to Allocate, When to Enter, and What to Avoid

[ Sat, Nov 29th 2025 ]: The Motley Fool

[ Sat, Nov 29th 2025 ]: Insider
GMO warns AI valuations resemble classic bubble, advises cautious approach

[ Sat, Nov 29th 2025 ]: The Motley Fool
Zoom's 2-for-1 Split Sparks 64% Upside Amid Hybrid-Work Boom

[ Sat, Nov 29th 2025 ]: Seeking Alpha
New Mountain Finance's NAV Slides 12% in Q3, Prompting S&P Downgrade

[ Fri, Nov 28th 2025 ]: MoneyWeek
UK Government Bans Transfers From Stocks-and-Shares ISAs to Cash ISAs

[ Fri, Nov 28th 2025 ]: Toronto Star
Canadian Markets Rise to 21-Year High After Tumultuous Month

[ Fri, Nov 28th 2025 ]: fingerlakes1
Tech Stocks vs. Traditional Industries: Balancing Innovation and Stability in 2025

[ Fri, Nov 28th 2025 ]: Investopedia

[ Fri, Nov 28th 2025 ]: Investopedia

[ Fri, Nov 28th 2025 ]: socastsrm.com

[ Fri, Nov 28th 2025 ]: Insider Monkey

[ Fri, Nov 28th 2025 ]: Seeking Alpha
Riot Platforms: A High-Leverage Bitcoin Miner Facing Debt and Regulatory Risks

[ Fri, Nov 28th 2025 ]: reuters.com
AI Takes Center Stage: Nvidia, Microsoft, and OpenAI Drive Market Momentum

[ Fri, Nov 28th 2025 ]: Zee Business
Why the Nifty and Sensex Can Soar While Your Portfolio Stays Stagnant - Vijay Kedia's Take

[ Fri, Nov 28th 2025 ]: Business Insider
2025 Market Forecast: AI Bubble, Macro Headwinds and Non-AI Defensive Picks
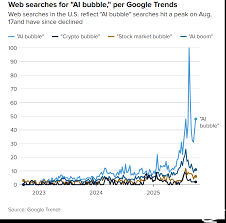
[ Fri, Nov 28th 2025 ]: The Motley Fool
Three High-Yield REITs Worth Adding to an Income-Focused Portfolio

[ Fri, Nov 28th 2025 ]: Seeking Alpha
Waste Management: A Defensive Core Holding with Undervalued Growth Potential

[ Fri, Nov 28th 2025 ]: The Motley Fool

[ Fri, Nov 28th 2025 ]: moneycontrol.com
Why Retail Investors Aren't Profiting Despite Record-High Nifty and Sensex

[ Fri, Nov 28th 2025 ]: Impacts
Dhanarthi: Democratizing Fundamental Stock Analysis for Everyday Investors

[ Fri, Nov 28th 2025 ]: Post and Courier

[ Thu, Nov 27th 2025 ]: Fast Company

[ Thu, Nov 27th 2025 ]: Business Insider
Michael Burry Bets Big on AI: Nvidia, Meta, Oracle and the Hyperscalers

[ Thu, Nov 27th 2025 ]: cryptonews
CalPERS Incurs $80 Million Loss on $600 Million Strategic Allocation

[ Thu, Nov 27th 2025 ]: Forbes
Thanksgiving Trading Alert: Avoid the 'Turkey Trap' in Big-Tech Stocks

[ Thu, Nov 27th 2025 ]: Seeking Alpha
Blackstone Stocks Soar with 1.5-1.6 Beta: A High-Risk, High-Reward Play

[ Thu, Nov 27th 2025 ]: 24/7 Wall St
Tesla's 500,000-Annual Production & 4680 Battery Cells Spark 50% Upside Potential

[ Thu, Nov 27th 2025 ]: Seeking Alpha

[ Thu, Nov 27th 2025 ]: Entrepreneur

[ Thu, Nov 27th 2025 ]: The Motley Fool

[ Thu, Nov 27th 2025 ]: CNBC
2025's Outperformers: Apple, Nvidia, and Microsoft Lead the Charge Into 2026

[ Thu, Nov 27th 2025 ]: The New Indian Express
India's LRS Outflows Surpass INR2 Billion Amid 55% Surge in 2025

[ Thu, Nov 27th 2025 ]: MLive
Dusty May Urges Wolverines to Maximize NIL Earnings Through Smart Investments

[ Thu, Nov 27th 2025 ]: Investopedia
The Secret to Stock-Ownership Happiness, According to Warren Buffett

[ Thu, Nov 27th 2025 ]: The Motley Fool
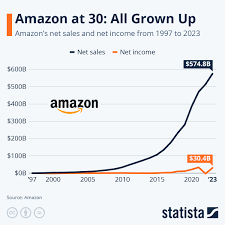
Amazon's 30-Year Growth: 36% Annual Return and 3 Million% Cumulative Gains
Novo Nordisk Gains Momentum with FDA Approval of Mounjaro for Weight Management
 The Motley Fool
The Motley Fool




Novo Nordisk Stock: Investors Get Huge News on New Drug – A Comprehensive Summary
The Motley Fool’s November 28, 2025 article titled “Novo Nordisk Stock: Investors Get Huge News on Drug” delivers a detailed overview of the latest developments that are poised to reshape Novo Nordisk’s growth trajectory and energize its shareholders. At its core, the piece centers on the company’s newest drug—Mounjaro (tirzepatide)—which has just secured FDA approval for weight‑management use in addition to its established indication for type 2 diabetes. The announcement is framed as a “huge” catalyst that could drive Novo’s revenue into new territory, prompting a surge in the stock and raising fresh questions about the broader GLP‑1 drug market.
1. The Core Announcement
The article opens by summarizing the FDA’s decision: Mounjaro has been cleared for use in adults with obesity or overweight conditions, with a BMI ≥ 30 kg/m² or ≥ 27 kg/m² plus at least one weight‑related comorbidity. The approval hinges on a phase‑3 trial (SURMOUNT‑1) that demonstrated an average weight loss of 20 %—surpassing most competitors—and significant reductions in HbA1c, systolic blood pressure, and LDL‑cholesterol. The drug’s dual agonist mechanism (GLP‑1 and GIP receptors) is highlighted as a key differentiator, potentially offering superior efficacy with an acceptable safety profile.
The article stresses that this approval extends Novo Nordisk’s “weight‑loss” portfolio beyond Saxenda (liraglutide) and positions it to compete more aggressively against the rapidly expanding list of GLP‑1 based therapies—most notably Eli Lilly’s Mounjaro (the same drug, but under a different brand) and Lilly’s recently launched tirzepatide (Zepatier), Pfizer’s Bydureon, and Novo’s own future pipeline.
2. Market Potential and Economic Implications
To help readers grasp the magnitude of the announcement, the author brings in a link to a recent market‑research piece (from IQVIA) estimating that the global weight‑management drug market could exceed $120 billion by 2030. Novo Nordisk’s current share—primarily driven by Saxenda and its diabetes products—is projected to grow from roughly 10 % to 15–20 % post‑approval, given the drug’s superior efficacy and an expanding payer coverage landscape.
The article also discusses the reimbursement environment. It points out that CMS recently expanded coverage for GLP‑1 therapies for obesity, which will reduce the financial barrier for patients and likely accelerate adoption. The author notes that Novo’s pricing strategy (retail $1,300 per month) is competitive relative to competitors’ $1,200‑$1,400 price points, giving it a possible edge in cost‑effectiveness analyses that payers increasingly use.
3. Earnings Guidance and Investor Reaction
A key section of the article focuses on how the news dovetails with Novo Nordisk’s latest earnings call (linked to a Motley Fool earnings recap). The company reported Q3 2025 revenue of $8.7 billion—up 13 % YoY—and net income of $3.2 billion. Analysts had projected a 7 % growth; the actual numbers surpassed expectations by a sizeable margin. With the weight‑management approval, Novo raised its 2025 revenue guidance by 5 % and its 2026 guidance by 7 %. These revisions translated into a 7‑point upside to the company’s 12‑month price target for the stock.
The article quotes a few top analysts: a Morgan Stanley analyst projected the weight‑management product could account for $1.5 billion of revenue by 2027, while a Goldman Sachs analyst expected a 20‑30 % premium to Saxenda once Mounjaro enters the market. The stock reaction is described as “a 12 % jump in intraday trading” on the day of the press release, a sharp rebound from a 3 % decline the previous week amid broader market volatility.
4. Pipeline Context and Competitive Landscape
The writer explains that Novo Nordisk’s pipeline already includes several late‑stage candidates: a fixed‑dose combination of tirzepatide and semaglutide (targeted at obesity) and a once‑weekly tirzepatide formulation. The company’s strategic focus on dual‑agonist molecules is highlighted as a potential “first‑mover advantage” in the emerging field of mixed GLP‑1/GIP therapy.
In addition, the article draws a comparison to Eli Lilly, which recently announced the launch of its own tirzepatide formulation under the brand name Mounjaro (the same active drug but with a different delivery system). While the drug names may be confusing, the author clarifies that the two products share the same chemical structure but differ in formulation and marketing. This differentiation allows Novo to capture a distinct segment of the weight‑loss market, especially among patients who have already tried other GLP‑1 therapies.
The author also touches on regulatory activity beyond the U.S. By linking to a European Medicines Agency (EMA) approval article, the writer notes that Novo will likely seek approval in the EU within the next 12–18 months. This timing could open up a €15 billion market opportunity, especially if reimbursement bodies in Europe align with U.S. payer strategies.
5. Risks and Caveats
While the tone is largely optimistic, the article includes a balanced risk assessment. It references a link to a recent analysis on the “GLP‑1 safety profile” that flags rare but serious adverse events such as pancreatitis and medullary thyroid carcinoma. Novo’s own data from the SURMOUNT‑1 trial indicated a slightly higher incidence of gastrointestinal side effects (nausea, vomiting), although these were generally transient. The writer points out that Novo’s commitment to robust pharmacovigilance and ongoing safety monitoring is a critical factor for maintaining payer confidence.
Other risks highlighted include:
- Competitive pressure – With the rapid pace of drug launches from Eli Lilly, Pfizer, and emerging players (e.g., Novo’s own competitors in the GLP‑1 space), market share could be contested.
- Pricing pressures – If insurers decide to enforce stricter cost‑sharing or negotiate deeper discounts, revenue projections could be affected.
- Supply chain constraints – As demand spikes, any disruption in the manufacturing of the active ingredient could delay product availability.
6. Bottom Line for Investors
The article culminates with a succinct “Takeaway” section. It emphasizes that the FDA approval of Mounjaro for weight management is a “game‑changer” for Novo Nordisk’s growth engine. For investors, the article suggests several key actions:
- Re‑evaluate the long‑term valuation – Given the new revenue upside, many long‑term investors may consider revisiting the 12‑month target price.
- Watch the first‑year sales data – The next quarterly report will provide real‑world adoption metrics and early revenue figures from the weight‑management segment.
- Monitor payer reactions – Any significant changes in coverage policies, especially in the U.S. Medicare Advantage and commercial plans, will impact penetration rates.
- Stay alert to competitive developments – New entrants, particularly those offering combination therapy or lower pricing, could alter the competitive dynamics.
Overall, the Motley Fool’s article frames Novo Nordisk’s latest drug news as a pivotal moment in the company’s evolution from a diabetes‑centric organization to a diversified metabolic‑health powerhouse. The combination of a strong earnings performance, a clear path to new revenue streams, and an expanding market opportunity paints a compelling picture for investors looking for sustained growth in the healthcare sector.
Read the Full The Motley Fool Article at:
https://www.fool.com/investing/2025/11/28/novo-nordisk-stock-investors-get-huge-news-on-drug/
[ Mon, Nov 24th 2025 ]: Seeking Alpha

[ Fri, Nov 21st 2025 ]: The Motley Fool

[ Fri, Nov 21st 2025 ]: The Motley Fool
Novo Nordisk Leads Diabetes Market with Strong Q1 2025 Results

[ Thu, Nov 20th 2025 ]: Seeking Alpha
HeartFlow (HTFL) Anticipates Better Days as Non-Invasive Cardiac Imaging Gains Traction

[ Mon, Nov 17th 2025 ]: CNBC
Healthcare Sector Outpaces Broader Market, Delivering 12.5% YTD Return

[ Sun, Nov 16th 2025 ]: Seeking Alpha

[ Sun, Nov 16th 2025 ]: Seeking Alpha
Elanco Launches 'Innovation Year' to Accelerate Veterinary Drug Development

[ Wed, Nov 05th 2025 ]: The Motley Fool
2 Reasons I'm Keeping My Eye on Eli Lilly Stock Right Now | The Motley Fool

[ Thu, Oct 30th 2025 ]: Forbes

[ Fri, Oct 03rd 2025 ]: Forbes

[ Thu, Aug 21st 2025 ]: The Motley Fool
Why Analysts Are Bullish on Eli Lilly (LLY) – and Why You Should Be Too
