by: Seeking Alpha
Celldex Announces Public Stock Offering to Fund Neurological & Immunological Therapies

Eli Lilly's Zepbound Approved: A New Era in Obesity Treatment





Wednesday, April 1st, 2026 - Eli Lilly's Zepbound (tirzepatide) has officially hit the market, following full FDA approval announced earlier today. The drug, an injectable aimed at combating obesity, represents a potential paradigm shift in how society addresses a growing public health crisis. While the scientific community largely hails the approval as a significant step forward, questions surrounding cost, accessibility, and manufacturing capacity loom large.
Zepbound's mechanism of action centers around mimicking the action of natural gut hormones - specifically, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). These hormones play a crucial role in regulating appetite and satiety. By mimicking their effects, Zepbound effectively helps individuals feel fuller for longer, leading to reduced caloric intake. The injectable formulation, delivered weekly, is similar to other increasingly popular weight-loss medications like Novo Nordisk's Wegovy and Ozempic.
The FDA's approval criteria target adults with a Body Mass Index (BMI) of 30 or higher, classifying them as obese. Crucially, the approval also extends to individuals with a BMI of 27 or greater who suffer from weight-related comorbidities - conditions exacerbated by excess weight, such as hypertension, type 2 diabetes, sleep apnea, and cardiovascular disease. This broader eligibility significantly expands the potential patient pool and acknowledges obesity as a complex medical condition, not simply a lifestyle choice.
A Blockbuster on the Horizon?
Analysts are predicting Zepbound will quickly become a blockbuster drug, potentially rivaling and even surpassing Wegovy in market share. Current projections estimate annual revenue exceeding $8 billion within the next five years, with some bullish forecasts reaching $15 billion. These numbers are driven by the sheer scale of the obesity epidemic. Over 40% of American adults are now considered obese, and the associated healthcare costs are astronomical. Zepbound offers a potentially effective treatment option where lifestyle interventions alone often fall short.
However, the promise of substantial revenue is tempered by concerns regarding cost and accessibility. While Eli Lilly has not yet publicly announced the final pricing, industry experts anticipate a price point comparable to, or potentially exceeding, Wegovy's current list price of around $1,300 per month. This high cost will likely place Zepbound out of reach for many who could benefit from it, especially those without comprehensive health insurance coverage. The debate over affordability and equitable access to obesity medications is expected to intensify.
Supply Chain Concerns Persist
Eli Lilly faces a significant challenge in scaling up manufacturing to meet anticipated demand. The experience of Novo Nordisk with Wegovy serves as a cautionary tale. Wegovy, approved in 2021, has been plagued by intermittent shortages due to manufacturing bottlenecks and supply chain disruptions. This led to frustration amongst patients and healthcare providers, and prompted Eli Lilly to proactively invest in expanding production facilities prior to Zepbound's launch.
Recent reports indicate that Eli Lilly has invested heavily in its manufacturing capabilities, including a significant expansion of its facility in Indiana and partnerships with contract manufacturing organizations. Despite these efforts, analysts remain cautiously optimistic about the company's ability to consistently meet demand, especially during the initial rollout phase. The use of complex biological processes in producing the drug necessitates stringent quality control, which can further slow down production.
Beyond the Pill: The Future of Obesity Treatment
The approval of Zepbound isn't just about a new medication; it's part of a broader shift in how obesity is understood and treated. There's increasing recognition that obesity is a chronic disease with complex biological underpinnings, requiring a multi-faceted approach. This includes lifestyle interventions (diet and exercise), behavioral therapy, and now, increasingly effective pharmacological treatments. Research is also underway to explore combination therapies and alternative delivery methods, such as oral formulations.
The long-term impact of Zepbound remains to be seen. Ongoing studies will focus on the drug's long-term efficacy and safety, as well as its potential impact on cardiovascular health and other obesity-related complications. The coming years will be crucial in determining whether Zepbound truly delivers on its promise to revolutionize obesity treatment and improve the lives of millions.
Read the Full CNBC Article at:
https://www.cnbc.com/2026/04/01/eli-lilly-secures-long-awaited-fda-approval-for-its-obesity-pill-heres-what-to-know.html
on: Tue, Feb 10th
by: The Motley Fool
Novo Nordisk Faces Production Hurdles Despite Wegovy Success

on: Tue, Feb 17th
by: The Motley Fool
Novo Nordisk's Cagrisema: A Potential Leap in Diabetes & Obesity Treatment
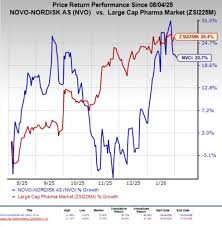
on: Fri, Feb 06th
by: CNBC

on: Thu, Mar 19th
by: CNBC

on: Thu, Feb 05th
by: The Motley Fool

on: Mon, Mar 23rd
by: Seeking Alpha
Novo Nordisk Stock Plunge Impacts Foundation, Raises R&D Concerns

on: Sun, Mar 08th
by: The Motley Fool

on: Tue, Feb 17th
by: Seeking Alpha

on: Fri, Jan 30th
by: The Motley Fool
FDA Delays Weight-Loss Drug Approval Due to Cardiovascular Concerns

on: Mon, Mar 23rd
by: The Motley Fool

on: Tue, Feb 17th
by: The Motley Fool
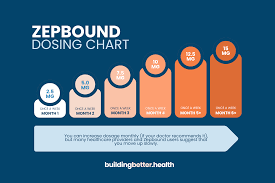
on: Tue, Mar 03rd
by: Seeking Alpha
Ascendis Pharma's Yuvjifel Gains EU Approval Amidst Legal Challenges









































