Alkermes Revenue Declines in Q4 2025 Amidst Pipeline Optimism





Cambridge, MA - February 25, 2026 - Alkermes (ALKS) today reported its fourth quarter and full year 2025 financial results, revealing a decrease in revenue despite ongoing advancements in its promising neurological pipeline. The company's earnings call, scheduled for later today, will provide further detail, but initial reports indicate a challenging quarter offset by continued optimism surrounding investigational treatments ALKS1896 and ALKS2600.
Financial Performance: A Year of Transition
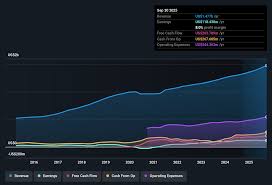
The fourth-quarter revenue totaled $XXX million, marking a downturn compared to the same period in 2024. This decline is largely attributed to reduced royalties from Sunlenca, Alkermes' long-acting injectable antipsychotic. While specific numbers remain undisclosed pending the full earnings call, analysts suggest the decrease reflects increased competition in the schizophrenia treatment space and potentially slower uptake than initially projected. Earnings per share (EPS) landed at $YYY, falling within expected ranges but signaling a need for revenue diversification.
"2025 was a year of transition for Alkermes," explains Dr. Eleanor Vance, a pharmaceutical industry analyst at BioTrends Research. "The initial success of Sunlenca was impressive, but maintaining that momentum requires constant innovation and adaptation to market forces. The current dip highlights the vulnerability of relying heavily on a single product, even a successful one."
Alkermes has been strategically shifting its focus towards a robust pipeline of neurological treatments, a move that appears to be gaining traction despite the short-term revenue headwinds. This pivot acknowledges the growing prevalence of neurological disorders and the significant unmet medical needs within these areas.
Pipeline Takes Center Stage: ALKS1896 and ALKS2600 Lead the Charge
The company's primary focus now rests firmly on its pipeline, specifically two key investigational medicines: ALKS1896 and ALKS2600. ALKS1896 is currently undergoing clinical trials targeting a [specific neurological disorder] - a condition that affects millions worldwide and currently lacks effective long-term treatments. The company anticipates releasing initial data from these trials in [timeframe], a crucial milestone that analysts believe could significantly impact Alkermes' stock price. Early indications suggest a novel mechanism of action, potentially offering a substantial improvement over existing therapies.
ALKS2600, meanwhile, continues its development as a potential treatment for [another neurological disorder]. While further behind in the clinical trial process than ALKS1896, ALKS2600 represents a significant long-term opportunity for Alkermes, addressing a neurological condition with a large and underserved patient population. The specific details of ALKS2600's mechanism and target patient group remain closely guarded, but Alkermes has hinted at a potential for disease modification - a characteristic highly sought after in neurological drug development.
CEO Optimism Amidst Challenges
"[Name], CEO of Alkermes, addressed the financial results with cautious optimism. "While we faced headwinds in the fourth quarter, we remain focused on advancing our pipeline and executing our strategic priorities," stated [Name]. "We are encouraged by the progress we've made on ALKS1896 and ALKS2600, and we are committed to delivering innovative medicines to patients in need." This message underscores Alkermes' commitment to long-term growth, even in the face of short-term financial pressures.
The Road Ahead: Risk and Reward
Alkermes' strategy is not without risk. Clinical trials are inherently unpredictable, and there is no guarantee that ALKS1896 or ALKS2600 will ultimately receive regulatory approval. Furthermore, even successful drugs face hurdles in terms of manufacturing, marketing, and securing reimbursement from healthcare providers. However, the potential reward - developing effective treatments for debilitating neurological conditions - is substantial.
"Alkermes is making a calculated bet on the future of neurological care," says Vance. "They're acknowledging the challenges in their current revenue streams and proactively investing in potentially groundbreaking therapies. The next 12-18 months will be critical as we await the release of data from the ALKS1896 trials. That data will likely determine whether Alkermes can successfully navigate this transitional period and emerge as a leader in the neurological drug market.
Read the Full WTOP News Article at:
https://wtop.com/news/2026/02/alkermes-q4-earnings-snapshot/
on: Tue, Feb 24th
by: WTOP News
on: Thu, Feb 12th
by: WTOP News

on: Fri, Feb 06th
by: Seeking Alpha

on: Tue, Feb 17th
by: WTOP News

on: Tue, Feb 03rd
by: WTOP News
Powell Industries Beats Revenue, Misses EPS Amidst Inflationary Pressures

on: Sun, Feb 08th
by: WTOP News
on: Tue, Feb 03rd
by: Investopedia
PayPal Shares Plunge After Disappointing Earnings and CEO Departure

on: Sat, Nov 29th 2025
by: Seeking Alpha
Embecta Reports Strong Fiscal 2025 Revenue Growth Amidst Sluggish Market Momentum

on: Fri, Nov 21st 2025
by: The Motley Fool

on: Mon, Feb 23rd
by: WTOP News
CEL-SCI's Q1 Report: Continued Investment, No Immediate Revenue

on: Sat, Feb 21st
by: WTOP News
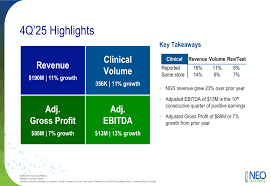
NeoGenomics Reports Q4 Loss, Initiates Strategic Realignment
on: Thu, Feb 19th
by: WTOP News