Seres Therapeutics Reports Q4 and Full-Year 2026 Results





Washington, D.C. - March 12, 2026 - Seres Therapeutics, a pioneering company at the forefront of microbiome therapeutics, released its fourth quarter and full year 2026 financial results today, painting a picture of cautious optimism amidst the inherent complexities of commercializing novel biological therapies. The report reveals increasing revenue, driven primarily by its existing product, SERBICA(R), but also underscores the substantial investment required to establish a foothold in this rapidly evolving field and, crucially, the continued operation at a loss.
While the specific percentage increase in revenue wasn't disclosed in initial reports, the growth indicates a strengthening market acceptance of microbiome-based interventions. SERBICA(R), currently approved for recurrent Clostridioides difficile infection (rCDI), is gaining traction, demonstrating the potential for targeted microbiome modulation to address serious health challenges. However, this growth hasn't yet translated into consistent profitability - a common hurdle for biotech companies focused on breakthrough therapies.
The company's substantial net loss for the quarter is largely attributed to the heavy investment in research and development (R&D). Developing microbiome therapeutics is a significantly complex undertaking. Unlike traditional pharmaceuticals, these therapies involve live bacterial consortia, requiring specialized manufacturing processes, stringent quality control, and a deeper understanding of the intricate interplay between the microbiome and the host immune system. Maintaining these complex systems and ensuring product stability add considerable cost.
The spotlight remains firmly on the ECHO-103 trial, a pivotal Phase 3 clinical study evaluating Seres' lead candidate for ulcerative colitis. This trial represents a critical inflection point for the company. Initial data released today continues to show promising signals, indicating potential efficacy in a disease area with significant unmet medical need. Ulcerative colitis, a chronic inflammatory bowel disease, affects millions globally, and current treatment options often fall short in providing lasting remission. The full ECHO-103 results, expected in the coming months, will be crucial for securing regulatory approval and, ultimately, driving revenue growth.
The Broader Promise of Microbiome Therapeutics & Seres' Position
Seres' commitment to pipeline expansion further illustrates the burgeoning potential of microbiome therapeutics. Beyond rCDI and ulcerative colitis, the company is actively exploring microbiome-based therapies for a diverse range of inflammatory and infectious diseases, including irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), and even potential applications in oncology. The core strategy centers around leveraging its proprietary microbial platform - a collection of carefully characterized bacterial strains and the expertise to cultivate and deliver them effectively.
The concept behind microbiome therapeutics is revolutionary. For decades, the microbiome - the trillions of bacteria, fungi, viruses, and other microorganisms residing in the human gut - was largely overlooked. However, recent advances in genomics and immunology have revealed its profound influence on virtually every aspect of human health, from digestion and immunity to mental well-being. Disruptions in the microbiome, known as dysbiosis, have been linked to a wide range of diseases.
Seres' approach involves reconstituting a healthy microbiome or modulating its composition to restore balance and alleviate disease symptoms. This differs significantly from traditional drug development, which often focuses on targeting specific molecules or pathways. Microbiome therapeutics aim to harness the power of the body's own microbial ecosystem to achieve therapeutic effects.
Challenges and the Path Forward
Despite the immense promise, the field faces significant challenges. The microbiome is incredibly complex and highly individualized, making it difficult to predict how a given therapy will respond in different patients. Manufacturing scalability, long-term stability of live bacterial products, and navigating regulatory pathways are all hurdles that companies like Seres must overcome.
"[Name], CEO of Seres Therapeutics, emphasized the company's commitment to navigating these challenges. "We are pleased with the revenue growth we've seen, reflecting increasing recognition of the value of microbiome therapeutics," [Name] stated. "However, we remain focused on executing our clinical development programs, particularly ECHO-103, which represents a significant opportunity to address a major unmet need in ulcerative colitis. We also continue to explore opportunities to expand our pipeline and leverage our microbiome platform."
Analysts are closely monitoring Seres' ability to achieve profitability and demonstrate the long-term clinical and commercial viability of its microbiome-based approach. The successful launch of SERBICA(R) provides a foundation, but the ECHO-103 trial remains the key catalyst for future growth. If approved, a therapy for ulcerative colitis would significantly expand Seres' addressable market and solidify its position as a leader in the rapidly evolving field of microbiome therapeutics. The coming months will be critical as the company works to unlock the full potential of this promising area of medicine and deliver value to patients and shareholders alike.
Read the Full WTOP News Article at:
https://wtop.com/news/2026/03/seres-therapeutics-q4-earnings-snapshot/
on: Wed, Mar 11th
by: WTOP News

on: Thu, Feb 12th
by: WTOP News

on: Tue, Feb 24th
by: WTOP News
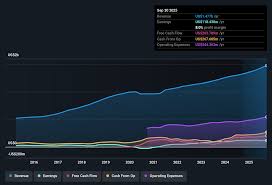
on: Sat, Feb 21st
by: WTOP News
NeoGenomics Reports Q4 Loss, Initiates Strategic Realignment
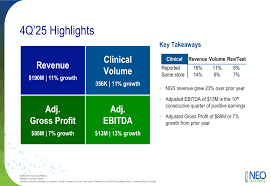
on: Tue, Feb 10th
by: WTOP News

on: Wed, Mar 11th
by: WTOP News

on: Tue, Mar 03rd
by: WTOP News

on: Fri, Feb 27th
by: WTOP News

on: Thu, Feb 26th
by: WTOP News
Alphatec Exceeds Expectations, Reports Strong Financial Results

on: Mon, Feb 23rd
by: WTOP News
CEL-SCI's Q1 Report: Continued Investment, No Immediate Revenue

on: Sat, Feb 07th
by: WTOP News

on: Thu, Feb 05th
by: The Motley Fool
















































